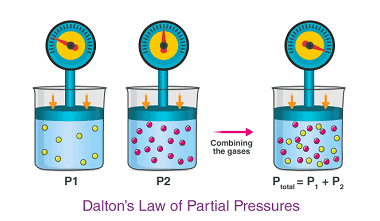
Raoult’s law
Raoult’s law is a law of thermodynamics that relates the vapor pressure of a component in a solution to its mole fraction. It states that the partial pressure of a component in a solution is equal to the product of the vapor pressure of the pure component and its mole fraction in the solution. Mathematically,…