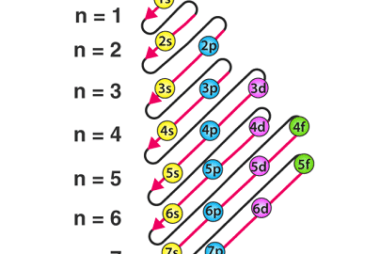
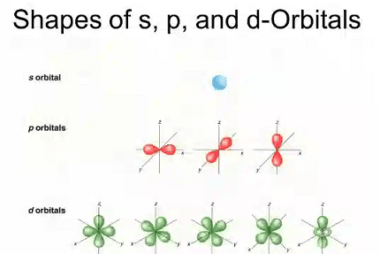
Second law of thermodynamics
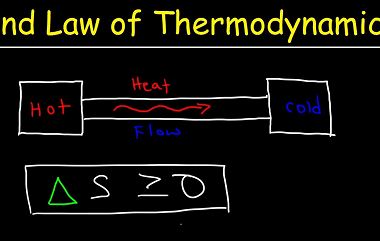
The second law of thermodynamics is a fundamental principle in physics that states that in any spontaneous process, the total entropy of a closed system will always increase over time, or remain constant in ideal cases. Entropy is a measure of the disorder or randomness of a system, and the second law of thermodynamics tells…