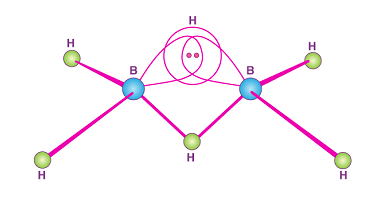
Group 15 Reactivity towards hydrogen
Group 15 elements, also known as pnictogens, include nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), and bismuth (Bi). These elements have five valence electrons in their outermost shell and tend to gain three electrons to achieve a stable octet configuration. In general, Group 15 elements do not react directly with hydrogen gas (H2) under…