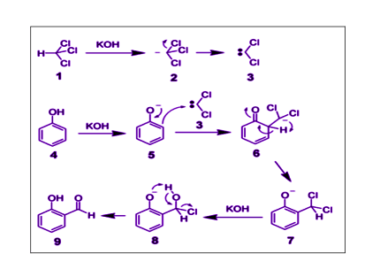
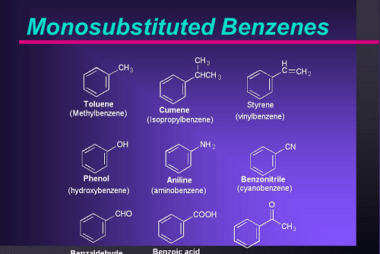
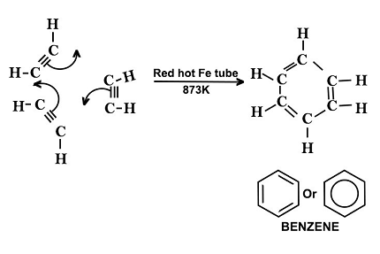
Electrophilic substitution reactions of phenol
Phenol undergoes electrophilic substitution reactions due to the presence of the electron-rich aromatic ring and the electron-withdrawing effect of the hydroxyl group. Here are some examples of electrophilic substitution reactions of phenol: What is Required Electrophilic substitution reactions of phenol For electrophilic substitution reactions of phenol to occur, the following conditions are required: Overall, electrophilic…