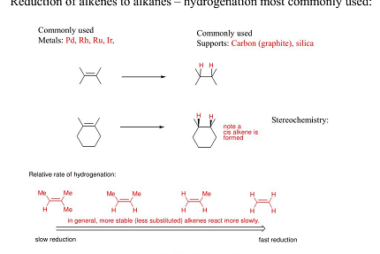
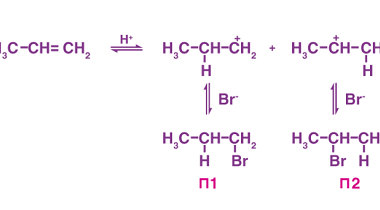
Reduction of Alkenes and Alkynes
Reduction of alkenes and alkynes involves the addition of hydrogen atoms to the carbon-carbon double or triple bond, respectively. This process is known as hydrogenation and can be accomplished through several methods. One common method is catalytic hydrogenation, which involves the use of a catalyst, such as platinum or palladium, to promote the reaction. The…