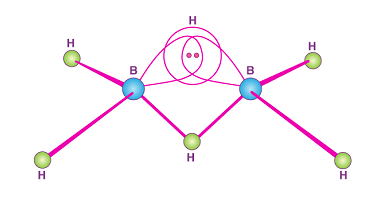
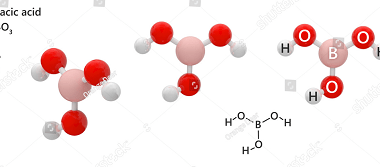
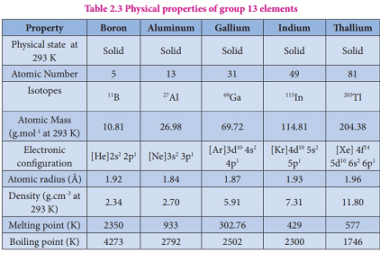
Group 13 Alums
Group 13 of the periodic table consists of the elements boron (B), aluminum (Al), gallium (Ga), indium (In), and thallium (Tl). These elements are collectively known as the boron group or the aluminum group. Aluminum and some of the other elements in Group 13 can form alums, which are double sulfates that have the general…