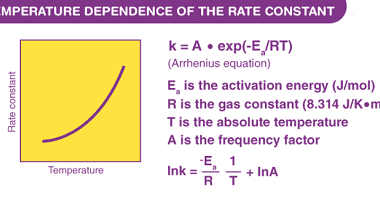
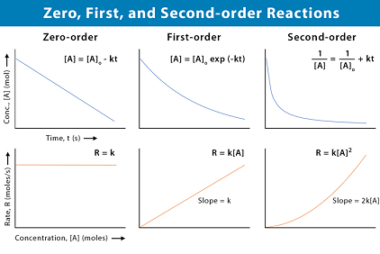
Henry’s law
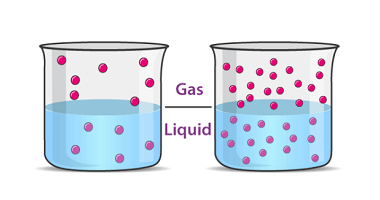
Henry’s law states that at a constant temperature, the amount of gas that dissolves in a liquid is directly proportional to the partial pressure of that gas in equilibrium with the liquid. In other words, the solubility of a gas in a liquid is directly proportional to the pressure of that gas above the liquid.…