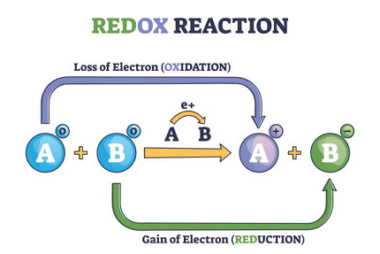
Integrated Course AIIMS-SYLLABUS Chemistry syllabus Redox reaction
Redox reactions Short for oxidation-reduction reactions, are chemical reactions in which there is a transfer of electrons between species. In these reactions, one species undergoes oxidation (loses electrons) while another species undergoes reduction (gains electrons). This transfer of electrons is accompanied by changes in the oxidation numbers of the elements involved. Key concepts related to…