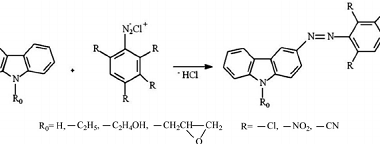
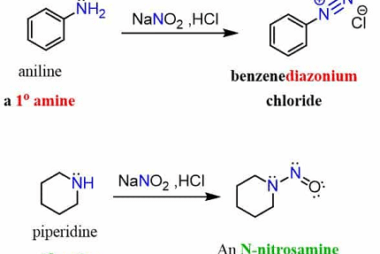
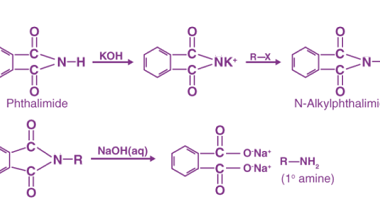
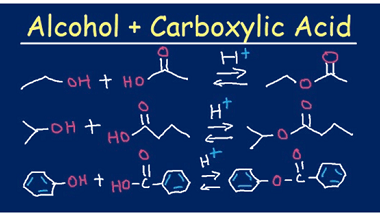
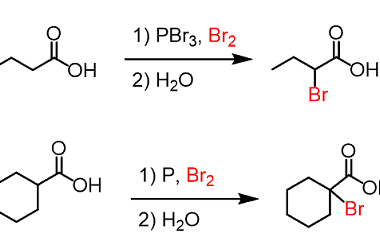
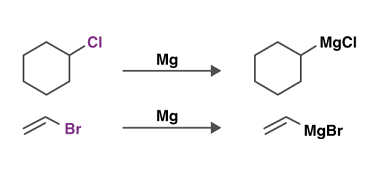
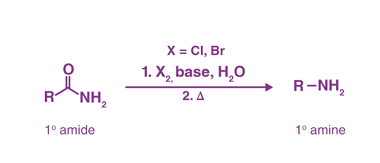Azo coupling reaction of diazonium salts of aromatic amines
Azo coupling reaction is a type of organic reaction that involves the reaction of a diazonium salt of an aromatic amine with another aromatic compound that contains an electron-rich group, such as an -OH or -NH2 group. The reaction results in the formation of an azo compound, which contains a -N=N- group. The general reaction…