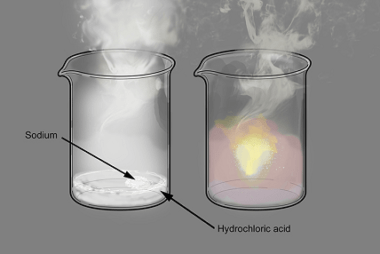
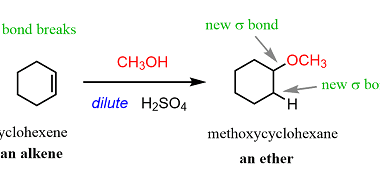
Nitriles
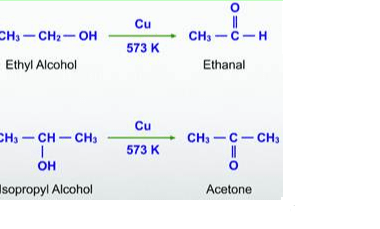
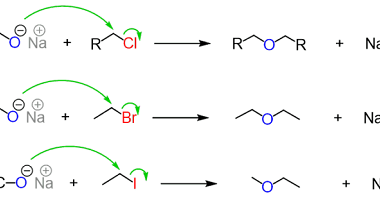
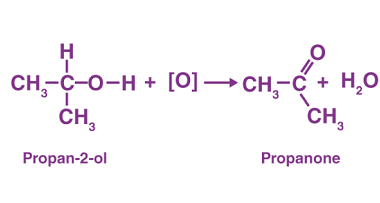
Aldehydes and ketones are organic compounds that contain a carbonyl group, which is a carbon atom double-bonded to an oxygen atom. In aldehydes, the carbonyl group is at the end of a carbon chain, whereas in ketones, it is in the middle. Nitriles, also known as cyano compounds, are organic compounds that contain a triple…