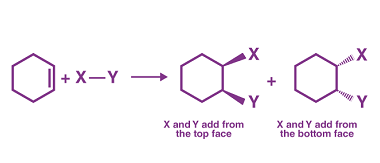
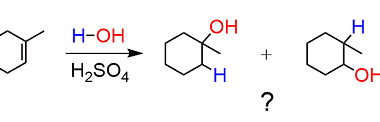
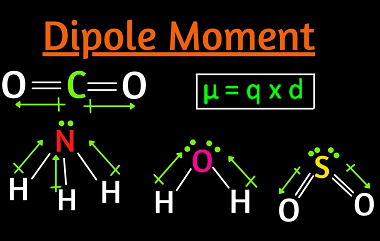
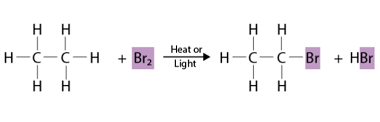
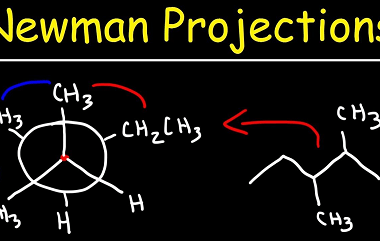
Elimination
Elimination is the process of removing or getting rid of something or someone. It can refer to various contexts, such as: What is Required Alkenes and Alkynes Elimination Required alkenes and alkynes elimination refers to a type of organic reaction known as an elimination reaction, in which a molecule loses a small molecule, such as…