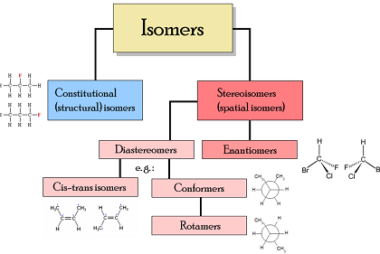
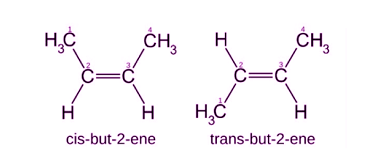
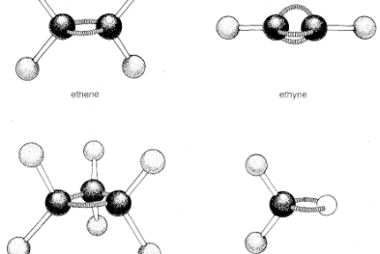
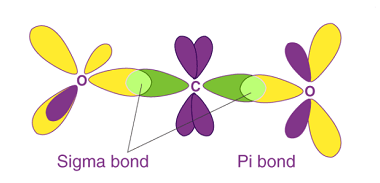
Stereoisomers and Stereochemical relationship
Stereoisomers are molecules that have the same molecular formula and connectivity of atoms but differ in the spatial arrangement of their atoms in three-dimensional space. They arise due to the presence of chiral centers, double bonds, or other forms of isomerism. Stereoisomers can be divided into two categories: enantiomers and diastereomers. Enantiomers are stereoisomers that…