Hydrolysis of disaccharides
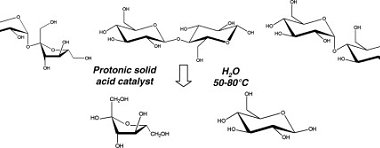
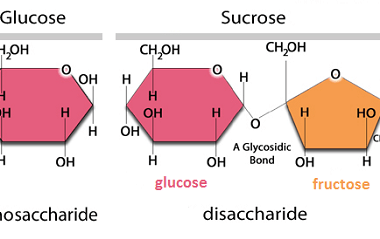
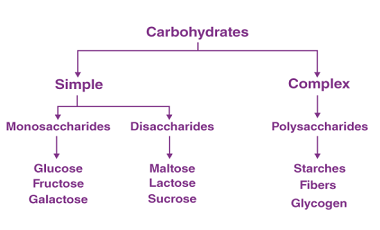
Hydrolysis of disaccharides is a chemical reaction that breaks down disaccharides into their constituent monosaccharides through the addition of water. Disaccharides are composed of two monosaccharides linked together by a glycosidic bond, which can be broken by the addition of water in the presence of an acid or enzyme. For example, the disaccharide sucrose (table…