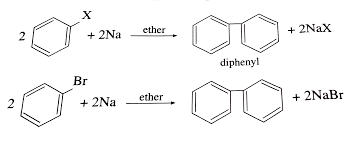
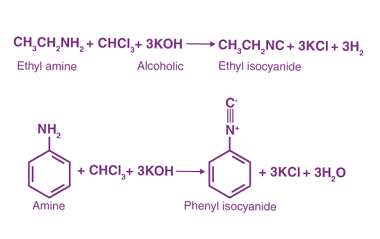
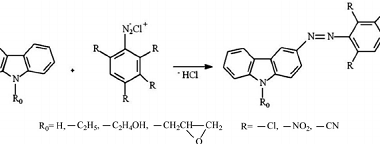
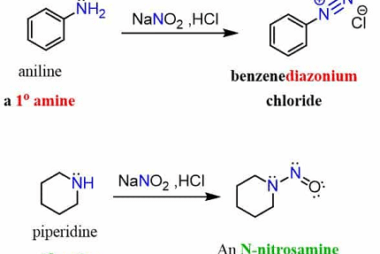
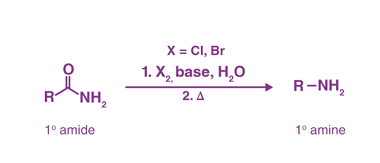Reactions: Fittig
Fittig is a reaction in organic chemistry named after the German chemist Friedrich Fittig. The Fittig reaction involves the coupling of two aryl or vinyl halides to form a biaryl or bivinyl compound, respectively, in the presence of a metallic sodium or potassium. The reaction is carried out in an inert solvent such as ether…