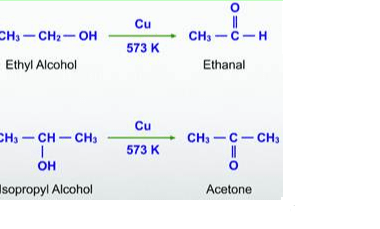
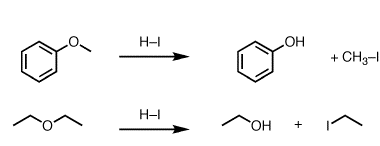
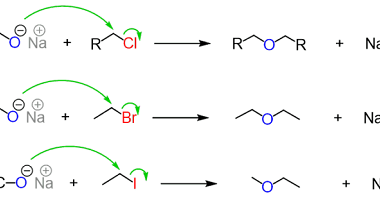
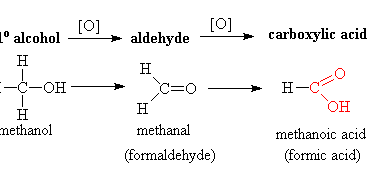
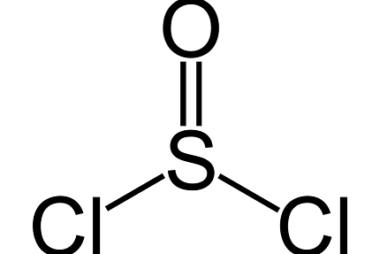
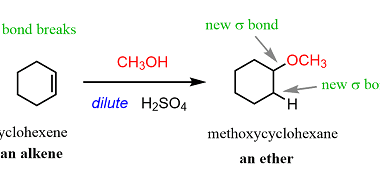
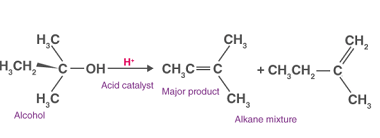
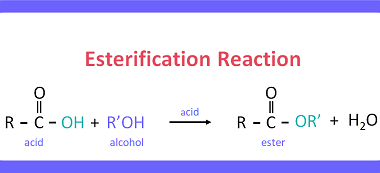
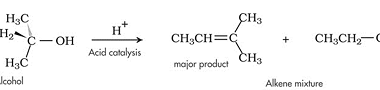
Preparation of: Aldehydes
Aldehydes can be prepared by several methods. Some of the commonly used methods are: These are some of the methods used for the preparation of aldehydes. What is Required Preparation of: Aldehydes The required preparation of aldehydes depends on the method being used for their synthesis. However, some common laboratory equipment and reagents that may…