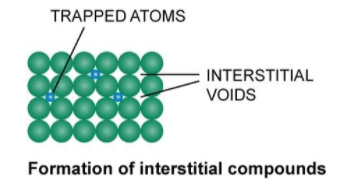
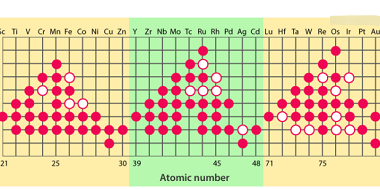
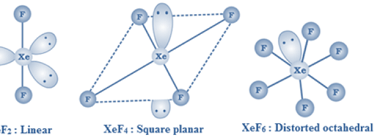
Alloys
An alloy is a combination of two or more metals, or a metal and another element, that results in a material with improved properties such as increased strength, hardness, or resistance to corrosion. Alloys are usually formed by mixing molten metals together and allowing them to cool and solidify. The resulting material has a unique…