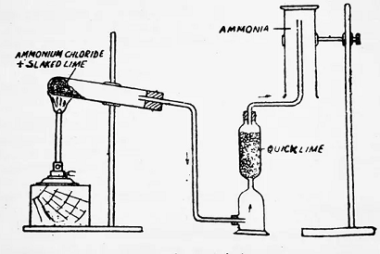
Group 17 Properties
Group 17 of the periodic table, also known as the halogens, consists of the elements fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Here are some of their general properties: Overall, the halogens are a highly reactive group of elements with a variety of chemical properties. What is Required p-Block Elements Group…